|
6/30/2023 0 Comments Nh3 acid or baseSo we have only the concentration of acetate to worry about here. Of CH3COOH times the concentration of hydroxide, so times the concentration of OH- this is all: over theĬoncentration of our reactants, and once again, we ignore water. So we write: Kb is equal to concentration of our products over concentration of our reactives. Alright, next we write ourĮquilibrium expression, and since this is acetateįunctioning as a base, we would write "Kb" here The concentration ofĪcetic acid would be X. We're assuming everything comes through equilibrium, here. So, at equilibrium, theĬoncentration of acetate would be. We'll be gaining X, aĬoncentration for the hydroxide. X over here, alright? And it's the same thing for hydroxide. We're going to lose X, and we're going to gain So if we make the concentration of the acetate anion, X, that reacts. Lose for the acetate anion, we gain for acetic acid. So CH3COO-, the acetateĪnion, when it reacts, is gonna turn into:ĬH3COOH, or acetic acid. Nothing has reacted, we should have a zero concentration for both of our products, right? So a zero concentrationįor our two products. And so that's the sameĬoncentration of our acetate anion, here, so we're gonna write: 0.25 molar, for the initial concentration of the acetate anion. 25 molar concentration of sodium acetate. Alright, so let's go ahead and write our initial concentrations here. And if you take a proton away from water, if you take an H+ away from H2O, you get OH-, or the hydroxide ion. 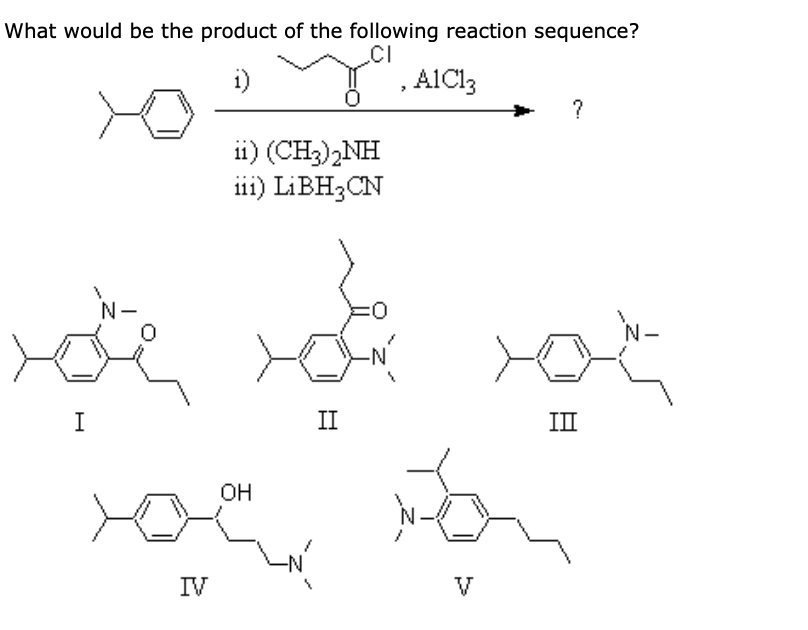
So if you add an H+ toĬH3COO-, you get CH3COOH. Going to react with water, and it's gonna function as a base: it's going to take a proton from water.  
So the acetate anion is theĬonjugate base to acetic acid. Going to react with water, but the acetate anions will. Have sodium ions, Na+, and acetate anions, CH3COO-, and the sodium cations aren't Of different salt solutions, and we'll start with this
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
